Home > Summary & Key Points
Summary & Key Points
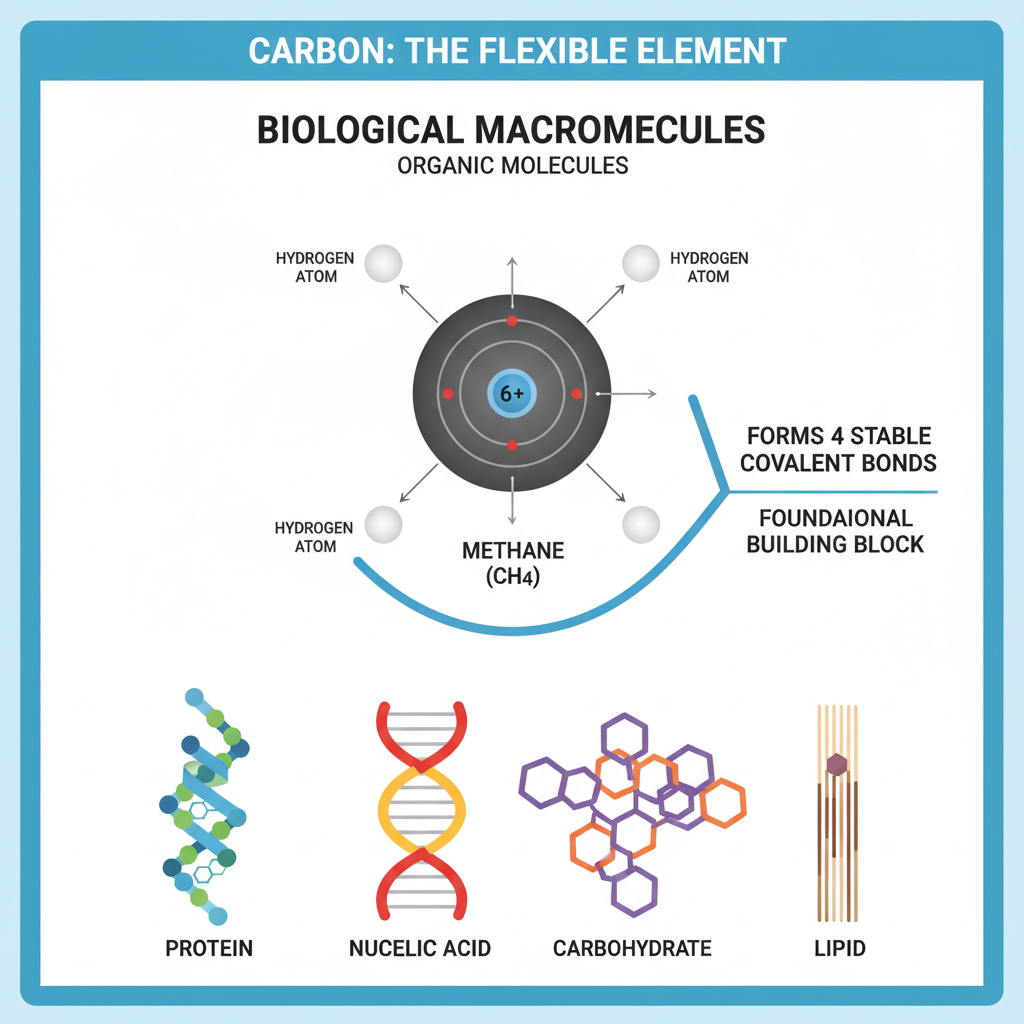
The document, “Introduction to Carbon’s Role and Bonding in Biological Molecules,” provides a foundational overview of why carbon is essential for life and how its unique atomic properties lead to the diverse structures of organic molecules.
Main Topics Summarized:
The text begins by establishing carbon as the foundational building block for all biological macromolecules (proteins, nucleic acids, carbohydrates, lipids), defining them as organic molecules due to their carbon content. It then delves into the carbon atom’s structure, explaining its atomic number (6), electron distribution (four valence electrons), and its ability to form four stable covalent bonds to satisfy the octet rule, making it a “flexible element.” Methane (CH4) is presented as a prime example of carbon’s tetrahedral bonding.
Next, the document introduces hydrocarbons, organic molecules made exclusively of carbon and hydrogen, highlighting their capacity to store large amounts of energy in their covalent bonds. It details how hydrocarbons can exist as various chains (straight, branched) and how the type of carbon-carbon bond (single, double, or triple) dictates the molecule’s three-dimensional shape or conformation. Single bonds allow rotation and lead to tetrahedral geometry, double bonds result in planar shapes with restricted rotation, and triple bonds lead to linear structures.
Finally, the text explores hydrocarbon rings, distinguishing between aliphatic hydrocarbons (linear chains or rings with only single bonds, like cyclohexane) and aromatic hydrocarbons (closed rings with alternating single and double bonds, exemplified by benzene). It notes the presence of these ring structures in important biological molecules such as amino acids, cholesterol, and hormones, while also mentioning the carcinogenicity of benzene.
Top 5 Key Takeaways:
- Carbon’s Fundamental Role in Life: Carbon is the unique and essential backbone for all organic molecules, including the complex macromolecules (proteins, DNA, carbohydrates, lipids) that constitute living organisms, enabling the vast diversity of life.
- Versatile Bonding Capacity: Due to its atomic number of 6 and four valence electrons, carbon can form up to four stable covalent bonds, satisfying the octet rule. This allows it to create diverse and complex structures by bonding with other carbons and various elements.
- Molecular Geometry Dictated by Bond Type: The specific type of covalent bond between carbon atoms significantly determines a molecule’s shape and flexibility: single bonds lead to tetrahedral geometry and allow rotation, double bonds result in a rigid planar shape, and triple bonds create a linear structure.
- Hydrocarbons as Energy-Rich Skeletons: Hydrocarbons, composed solely of carbon and hydrogen, serve as the basic structural framework for biological molecules. The numerous covalent bonds within hydrocarbons store substantial chemical energy, released upon oxidation.
- Structural Diversity: Chains and Rings: Carbon atoms can link together in various forms, including straight, branched, or cyclic (ring) chains. These rings are further classified as aliphatic (single bonds) or aromatic (alternating single and double bonds, like benzene), both of which are integral components of numerous biological compounds.
Related Videos to Explore
For the point: Versatile Bonding Capacity: Due to its atomic number of 6 and four valence electrons, carbon can form up to four stable covalent bonds, satisfying the octet rule. This allows it to create diverse and complex structures by bonding with other carbons and various elements.
- Carbon: The Stuff of Life
- Carbon and its Compounds - Introduction | Don’t Memorise
- The Carbon Atom and Bonding (Organic Chemistry Basics) - The Organic Chemistry Tutor
For the point: Molecular Geometry Dictated by Bond Type: The specific type of covalent bond between carbon atoms significantly determines a molecule’s shape and flexibility: single bonds lead to tetrahedral geometry and allow rotation, double bonds result in a rigid planar shape, and triple bonds create a linear structure.
- The Chemistry of Carbon-Carbon Double Bonds: Ethene and Beyond
- How Do Single, Double, And Triple Bonds Affect Bond Angles?
- Do Single, Double, Or Triple Bonds Change Bond Angles Differently?
See also: 02_Outline, 04_Glossary